Post Market Insights
Our real-world evidence solution to your wound care PMS challenge
Under EU Medical Device Regulation (MDR 2017/745) (Annex XIV, Part B), manufacturers are required to operate a continuous Post-Market Surveillance (PMS) system for every device. This is not a static compliance exercise, but an ongoing, risk-proportionate process to ensure clinical performance and safety remain robust throughout the product lifecycle.
Today, PMS has evolved into far more than a regulatory requirement. It is a critical commercial lever.
Across the UK and EU, healthcare systems are shifting towards real-world evidence (RWE) and value-based procurement, where purchasing and reimbursement decisions are increasingly driven by demonstrable patient outcomes, system efficiencies, and cost-effectiveness in everyday clinical practice.
In parallel, frameworks such as the UK Drug Tariff Part IX review are raising the bar for evidence required to secure and retain reimbursement. Manufacturers must now go beyond initial clinical data, generating ongoing, real-world insights that clearly demonstrate value in use, not just efficacy in controlled settings.
The implication is clear: post-market insight now underpins both market access and market retention. Without a structured approach to real-world data generation, even established products risk losing competitive position.
How MDS Can Help
MDS designs and delivers PMS programmes that align regulatory compliance with commercial strategy. Our UK-based post-market studies are built to generate meaningful real-world evidence, supporting not only MDR obligations, but also value-based procurement, reimbursement retention, and long-term market differentiation.

Frequency of PSUR Submission
- Class IIa and IIb devices: A PSUR must be updated at least every two years.
- Class III and implantable devices: The PSUR must be updated and submitted to the Notified Body annually.
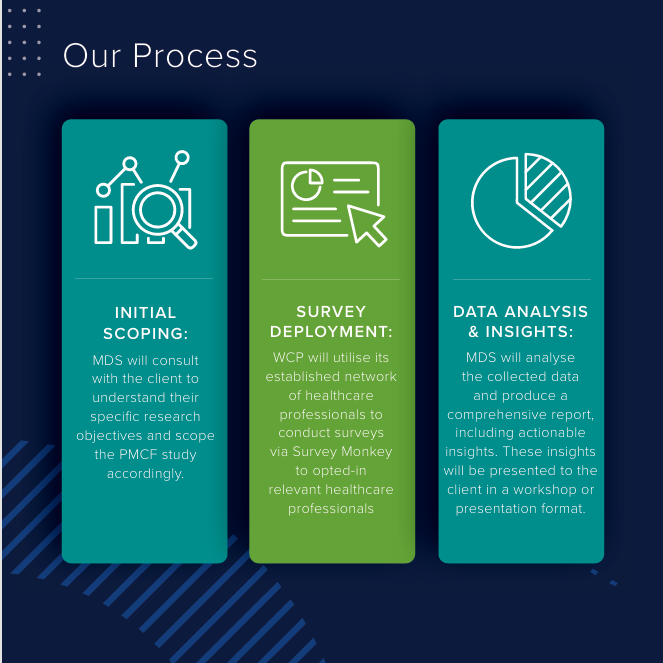
Our Solution:
To overcome these challenges, Med Dev Services has partnered with Wound Care People, a leading medical education provider in the wound care sector, to introduce Post Market Insights™.
MDS has exclusive access to a database of over 135,000 active, GDPR compliant, UK healthcare professionals who can provide real-world feedback on your device.
Post Market Surveillance Feedback for Wound Dressings
The Evidence Challenge:
Whilst in-house Regulatory & QA Departments are well versed in handling the constant stream of MDR hurdles facing them, one key problem still remains, collecting product relevant feedback from the professionals that use their devices.
Complex Supply Chains
- Indirect Distribution Channels: Wound dressings are often sold through wholesalers and distributors before reaching the clinic.
- Fragmented Feedback Sources: Feedback can come from multiple, non-standardised sources, including hospitals, clinics and home care services.
Limited Direct Clinician Engagement
- Lack of Direct Access to Clinicians: Regulatory teams often have limited direct engagement with the clinicians using the products, frequently the task is delegated to the not always co-operative sales team.
- Feedback Delays: Clinicians may only report issues when there is a significant problem, resulting in a lack of real-time feedback in use.
Regulatory Burden and Compliance Requirements
- High Volume of Data: Regulatory requirements such as the MDR necessitate continuous and comprehensive data collection. Keeping up with these requirements, particularly for high-risk devices, can be overwhelming for manufacturers.
- Data Integration: Compiling data from diverse sources into a format that meets the stringent requirements for post-market surveillance can be time-consuming and resource-intensive.
- Data Privacy Concerns: Navigating data protection regulations (GDPR) may also complicate the collection and sharing of patient-specific feedback.
Targeted feedback on your device
Post Market Insights™
- Designed specifically to meet the regulatory demands of MDR audits
- Helps collect, analyse and report clinical data for regulatory submissions
- Ensures legal manufacturers are audit-ready and compliant with PMS and PMCF requirements
